How Do You Know That Dichromate Has Been Reduced
| CHROMIUM This page looks at some aspects of chromium chemistry required for United kingdom A level (and its equivalents). It includes: reactions of chromium(III) ions in solution (summarised from elsewhere on the site); the interconversion of the various oxidation states of chromium; the chromate(VI)-dichromate(Six) equilibrium; and the apply of dichromate(Vi) ions as an oxidising agent (including titrations). The commencement part of this page is a summary of the reactions of chromium(3) ions in solution. You lot will find links to other pages where these reactions are discussed in more detail. You are very unlikely to need everything on this page. Check your syllabus and past papers to discover out exactly what you need to know. Reactions of chromium(Three) ions in solution The simplest ion that chromium forms in solution is the hexaaquachromium(3) ion - [Cr(H2O)vi]3+. | |
| Notation:If y'all aren't happy most circuitous ions (including the manner they are bonded and named), it would pay you to follow this link and explore the first couple of pages in the circuitous ions menu before you become on. Use the Dorsum button on your browser to return to this page. | |
| The acerbity of the hexaaqua ions In common with the other three+ ions, the hexaaquachromium(III) ion is fairly acidic - with a pH for typical solutions in the 2 - 3 range. The ion reacts with water molecules in the solution. A hydrogen ion is lost from one of the ligand water molecules: The complex ion is acting equally an acid past donating a hydrogen ion to h2o molecules in the solution. The water is, of form, interim every bit a base past accepting the hydrogen ion. Considering of the confusing presence of water from two different sources (the ligands and the solution), it is easier to simplify this: However, if you write information technology like this, remember that the hydrogen ion isn't just falling off the circuitous ion. It is beingness pulled off past a water molecule in the solution. Whenever y'all write "H+ (aq)" what you really hateful is a hydroxonium ion, HthreeO+. | |
| Note:You lot volition find the full reasons for the acidity of hexaaqua ions if you follow this link. You only need to read the outset of that page which concentrates on explaining the acidity of the hexaaquairon(III) ion. What is said applies equally to the chromium-containing ion. Use the BACK button on your browser to return to this page. | |
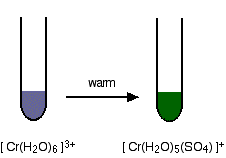
| Ligand exchange reactions involving chloride or sulphate ions The hexaaquachromium(Three) ion is a "difficult to describe" violet-blueish-grey color. Nevertheless, when it is produced during a reaction in a test tube, it is oftentimes light-green. We nigh e'er draw the dark-green ion as beingness Criii+ (aq) - implying the hexaaquachromium(III) ion. That's actually an over-simplification. What happens is that one or more than of the ligand water molecules get replaced past a negative ion in the solution - typically sulphate or chloride. Replacement of the water by sulphate ions You can do this simply by warming some chromium(3) sulphate solution. 1 of the h2o molecules is replaced by a sulphate ion. Notice the change in the accuse on the ion. 2 of the positive charges are cancelled by the presence of the ii negative charges on the sulphate ion. Replacement of the h2o by chloride ions In the presence of chloride ions (for example with chromium(Three) chloride), the most normally observed colour is green. This happens when two of the water molecules are replaced past chloride ions to give the tetraaquadichlorochromium(Three) ion - [Cr(H2O)4Cl2]+. In one case again, notice that replacing water molecules by chloride ions changes the charge on the ion. | |
| Note:You will notice an extensive give-and-take of ligand exchange reactions if you follow this link. Employ the BACK button on your browser to render to this page. | |
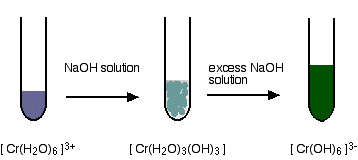
| Reactions of hexaaquachromium(III) ions with hydroxide ions Hydroxide ions (from, say, sodium hydroxide solution) remove hydrogen ions from the h2o ligands attached to the chromium ion. Once a hydrogen ion has been removed from three of the water molecules, you are left with a complex with no charge - a neutral circuitous. This is insoluble in h2o and a precipitate is formed. | |
| Notation:The colour coding is to testify that this isn't a ligand exchange reaction. The oxygens which were originally attached to the chromium are still fastened in the neutral complex. | |
| But the process doesn't stop there. More hydrogen ions are removed to give ions like [Cr(H2O)2(OH)4]- and [Cr(OH)6]3-. For example: The precipitate redissolves because these ions are soluble in h2o. In the test-tube, the colour changes are: | |
| Annotation:You will find the reactions between hexaaqua ions and hydroxide ions discussed in detail if yous follow this link. Use the Back push on your browser to render to this page. | |
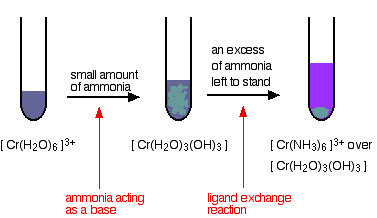
| Reactions of hexaaquachromium(3) ions with ammonia solution The ammonia acts every bit both a base and a ligand. With a small amount of ammonia, hydrogen ions are pulled off the hexaaqua ion exactly equally in the hydroxide ion example to give the same neutral complex. That precipitate dissolves to some extent if you lot add an excess of ammonia (especially if it is full-bodied). The ammonia replaces h2o every bit a ligand to give hexaamminechromium(III) ions. | |
| Note:You lot might wonder why this second equation is given starting from the original hexaaqua ion rather than the neutral complex. Explaining why the precipitate redissolves is quite complicated. You will find the explanation in full (although by reference to the corresponding copper case) on the page nigh the reactions between hexaaqua ions and ammonia solution. Utilise the Back button on your browser to render to this page. | |
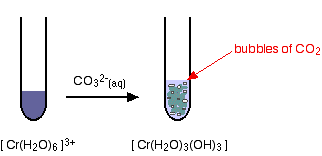
| The colour changes are: Reactions of hexaaquachromium(Three) ions with carbonate ions If y'all add sodium carbonate solution to a solution of hexaaquachromium(Iii) ions, you get exactly the aforementioned precipitate every bit if you added sodium hydroxide solution or ammonia solution. This time, it is the carbonate ions which remove hydrogen ions from the hexaaqua ion and produce the neutral complex. Depending on the proportions of carbonate ions to hexaaqua ions, you will get either hydrogencarbonate ions formed or carbon dioxide gas from the reaction betwixt the hydrogen ions and carbonate ions. The more ordinarily quoted equation shows the formation of carbon dioxide. Apart from the carbon dioxide, there is cypher new in this reaction: | |
| Note:You lot volition find the reactions between hexaaqua ions and carbonate ions discussed in detail if y'all follow this link. Employ the BACK push on your browser to return to this folio. | |
| The oxidation of chromium(III) to chromium(VI) An excess of sodium hydroxide solution is added to a solution of the hexaaquachromium(3) ions to produce a solution of greenish hexahydroxochromate(III) ions. This is then oxidised by warming it with hydrogen peroxide solution. You somewhen get a bright yellow solution containing chromate(Half-dozen) ions. The equation for the oxidation phase is: | |
| Note:Although it is yet a circuitous ion, you don't write square brackets effectually the chromate(Half dozen) ion - any more than yous would around a sulphate or carbonate ion. If you desire to know how to work out this equation , follow this link. Utilise the Dorsum push on your browser to return to this page. | |
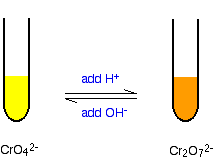
| Some chromium(VI) chemistry The chromate(VI)-dichromate(Half-dozen) equilibrium You lot are probably more familiar with the orangish dichromate(VI) ion, CrtwoOseven 2-, than the yellow chromate(Vi) ion, CrO4 2-. Changing between them is easy: If you add dilute sulphuric acid to the yellow solution it turns orange. If you add sodium hydroxide solution to the orange solution it turns yellow. | |
| Notation:If you had simply produced the yellow chromate(VI) ions past oxidising chromium(III) ions using hydrogen peroxide, yous tin't convert them into dichromate(VI) ions without taking a precaution first. In the presence of acid, dichromate(VI) ions react with any hydrogen peroxide which is left in the solution from the original reaction. To prevent this, you rut the solution for some time to decompose the hydrogen peroxide into water and oxygen earlier adding the acid. | |
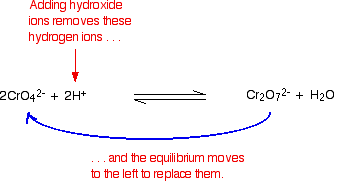
| The equilibrium reaction at the centre of the interconversion is: If you add extra hydrogen ions to this, the equilibrium shifts to the right. This is consequent with Le Chatelier's Principle. | |
| Note:If you aren't familiar with Le Chatelier's Principle, you should follow this link and read the first function of that page about the effect of concentration on position of equilibrium. Use the Back button on your browser to render to this page. | |
| If yous add together hydroxide ions, these react with the hydrogen ions. The equilibrium tips to the left to replace them. Making potassium dichromate(VI) crystals Potassium dichromate crystals can be made past a combination of the reactions we've already looked at on this folio. Starting from a source of chromium(III) ions such as chromium(III) chloride solution: You add together potassium hydroxide solution to give first a grey-greenish precipitate and and then the night green solution containing [Cr(OH)6]3- ions. This is all described in detail farther up the page. Notice that you lot have to use potassium hydroxide. If y'all used sodium hydroxide, you lot would end up eventually with sodium dichromate(Vi). Now you lot oxidise this solution by warming it with hydrogen peroxide solution. The solution turns yellow every bit potassium chromate(VI) is formed. This reaction is as well described farther upwards the page. All that is left is to convert the yellow potassium chromate(VI) solution into orangish potassium dichromate(VI) solution. You lot may remember that that is done past calculation acid. This is described to a higher place if you have forgotten. Unfortunately in that location is a problem here. Potassium dichromate will react with any excess hydrogen peroxide to give initially an unstable deep bluish solution and it eventually gives the original chromium(Iii) ions again! To get around this, you first need to destroy any excess hydrogen peroxide. This is done by boiling the solution. Hydrogen peroxide decomposes on heating to give h2o and oxygen. The solution is boiled until no more than bubbling of oxygen are produced. The solution is heated further to concentrate it, and then concentrated ethanoic acid is added to acidify it. Orange crystals of potassium dichromate are formed on cooling. The reduction of dichromate(VI) ions with zinc and an acrid Dichromate(Vi) ions (for example, in potassium dichromate(Half-dozen) solution) tin be reduced to chromium(3) ions and and then to chromium(2) ions using zinc and either dilute sulphuric acid or hydrochloric acid. Hydrogen is produced from a side reaction between the zinc and acid. This must be allowed to escape, only you need to keep air out of the reaction. Oxygen in the air rapidly re-oxidises chromium(2) to chromium(III). An like shooting fish in a barrel style of doing this is to put a bit of cotton wool in the top of the flask (or exam-tube) that you are using. This allows the hydrogen to escape, just stops most of the air getting in against the flow of the hydrogen. The reason for the inverted commas effectually the chromium(III) ion is that this is a simplification. The exact nature of the complex ion will depend on which acrid y'all utilize in the reduction process. This has already been discussed towards the top of the page. | |
| Note:To re-read this utilise this link. | |
| The equations for the two stages of the reaction are: For the reduction from +6 to +3: For the reduction from +3 to +2: | |
| Note:If y'all don't know how to work out equations like this, you can find out how to exercise information technology on the page near writing ionic equations for redox reactions. Use the BACK push button on your browser to return to this page. | |
| Using potassium dichromate(VI) as an oxidising agent in organic chemistry Potassium dichromate(VI) solution acidified with dilute sulphuric acid is commonly used as an oxidising agent in organic chemistry. It is a reasonably strong oxidising agent without beingness so powerful that it takes the whole of the organic molecule to pieces! (Potassium manganate(VII) solution has some tendency to practise that.) It is used to:
For example, with ethanol (a chief alcohol), you lot tin get either ethanal (an aldehyde) or ethanoic acid (a carboxylic acid) depending on the conditions.
In organic chemistry, these equations are oftentimes simplified to concentrate on what is happening to the organic molecules. For case, the last ii could be written: The oxygen written in square brackets just means "oxygen from an oxidising agent". | |
| Annotation:These are not a proper substitute for real equations. Simply use them if your examiners are happy with them. Check your syllabus and look at past papers and marker schemes. If you are working towards a United kingdom of great britain and northern ireland-based exam and don't accept these things, you lot can find out how to get agree of them past going to the syllabuses page. | |
| Using this same reaction to make chrome alum crystals You volition find chrome alum nether all sorts of unlike names:
. . . and various others! Yous volition also notice variations on its formula. For example:
The outset of these formulae is just the other ones divided by two and rearranged a chip. Personally, I prefer the second one because information technology is easier to empathise what is going on. Chrome alum is known equally a double salt . If y'all mix solutions of potassium sulphate and chromium(III) sulphate and so that their molar concentrations are the same, the solution behaves but like you would expect of such a mixture. It gives the reactions of chromium(Iii) ions, of potassium ions, and of sulphate ions. However, if you crystallise information technology, instead of getting mixed crystals of potassium sulphate and chromium(III) sulphate, the solution crystallises as single deep majestic crystals. These are "chrome alum". Chrome alum crystals can be made by reducing acidified potassium dichromate(VI) solution using ethanol, and then crystallising the resulting solution. Assuming you use an excess of ethanol, the primary organic product volition be ethanal - and nosotros've already seen this equation in a higher place: This ionic equation manifestly doesn't contain the spectator ions, potassium and sulphate. Feeding those back in gives the full equation: If you lot expect at the peak line on the right-hand side of the equation, you lot will encounter that the chromium(III) sulphate and potassium sulphate are produced in exactly the right proportions to make the double table salt. What you lot do, then, is this: | |
| Note:I am not giving quantities and exact weather - there are practical and condom considerations which brand me reluctant to do that. If you want precise details, they aren't hard to find. | |
| Yous first with a solution of potassium dichromate(Half dozen) to which has been added some concentrated sulphuric acid. The solution is so cooled past continuing it in ice. An excess of ethanol is added slowly with stirring so that the temperature doesn't ascent too much. | |
| Note:If the solution gets too warm, you get a ligand exchange reaction between h2o molecules attached to the chromium(III) ions produced and sulphate ions in the solution. This leads to the greenish class of chromium(Three) sulphate described higher upward the page. To make chrome alum crystals, you lot have to stop this happening. | |
| When all the ethanol has been added, the solution is left over-night, preferably in a refrigerator, to crystallise. The crystals can be separated from the remaining solution, washed with a piddling pure water and then dried with filter paper. Using potassium dichromate(Half-dozen) as an oxidising amanuensis in titrations Potassium dichromate(6) is often used to estimate the concentration of fe(Ii) ions in solution. Information technology serves as an culling to using potassium manganate(Seven) solution. | |
| Note:Potassium manganate(7) titrations are described fully on the page virtually manganese chemistry. | |
| In practice There are advantages and disadvantages in using potassium dichromate(VI). Advantages
Disadvantage
The calculation The one-half-equation for the dichromate(VI) ion is: . . . and for the atomic number 26(II) ions is: Combining these gives: You tin can run across that the reacting proportions are ane mole of dichromate(VI) ions to 6 moles of iron(II) ions. In one case you take established that, the titration adding is going to be only like any other one. | |
| Notation:If you aren't very good at doing titration calculations, you might be interested in my chemical science calculations volume. | |
| Testing for chromate(Six) ions in solution Typically, you lot would be looking at solutions containing sodium, potassium or ammonium chromate(VI). Most chromates are at all-time just slightly soluble; many we would count every bit insoluble. The bright yellow colour of a solution suggests that information technology would be worth testing for chromate(Six) ions. Testing by adding an acid If yous add some dilute sulphuric acid to a solution containing chromate(Vi) ions, the colour changes to the familiar orange of dichromate(Vi) ions. Y'all can't rely on this as a test for chromate(Half-dozen) ions, however. It might be that you have a solution containing an acrid-base indicator which happens to have the same colour alter! Testing by calculation barium chloride (or nitrate) solution Chromate(VI) ions will give a yellowish precipitate of barium chromate(Half-dozen). | |
 Annotation:The precipitate colour is very similar to the background colour I employ on Chemguide pages, which makes the last diagram a bit difficult to see. The photo on the correct is by courtesy of Professor Stanley G. Smith of the The Academy of Illinois at Urbana-Champaign. Annotation:The precipitate colour is very similar to the background colour I employ on Chemguide pages, which makes the last diagram a bit difficult to see. The photo on the correct is by courtesy of Professor Stanley G. Smith of the The Academy of Illinois at Urbana-Champaign. | |
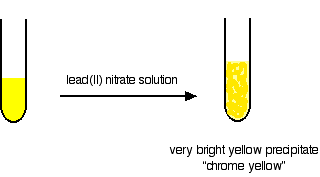
| Testing by adding lead(2) nitrate solution Chromate(Half dozen) ions will requite a bright yellowish precipitate of lead(II) chromate(Six). This is the original "chrome yellow" paint pigment. Questions to test your agreement If this is the beginning set of questions you have done, please read the introductory page earlier you start. You volition need to use the BACK Push button on your browser to come back here afterwards. questions on chromium answers
To the transition metal card . . . To the Inorganic Chemistry menu . . . To Main Carte du jour . . . © Jim Clark 2003 (last modified June 2015) | |
williamsthadmilly.blogspot.com
Source: https://www.chemguide.co.uk/inorganic/transition/chromium.html
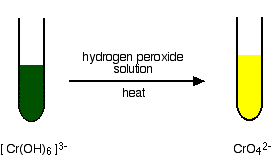




0 Response to "How Do You Know That Dichromate Has Been Reduced"
Post a Comment